Psychotropic polypharmacy is common, yet there has been limited guidance on when and how to discontinue medications. A 45-member international task force from the American Society of Clinical Psychopharmacology used a Delphi process to establish consensus recommendations on when deprescribing should be considered and how it should be approached.
Consensus was achieved on 44 of 50 statements (88%), with several clear themes emerging. Deprescribing should be considered when benefits are absent, partial, or outweighed by risks, but only after confirming adherence and adequate trial duration. The task force strongly emphasized routine, periodic medication review, shared decision-making, and addressing psychological factors like patient fears and expectations. Importantly, only one medication should be changed at a time, and deprescribing should include close follow-up and monitoring for relapse. Additional guidance highlighted the need to evaluate drug-drug interactions, avoid unnecessary polypharmacy (especially with mechanistically redundant agents), and prioritize deprescribing in cases of adverse effects, poor tolerability, or lack of clear indication.
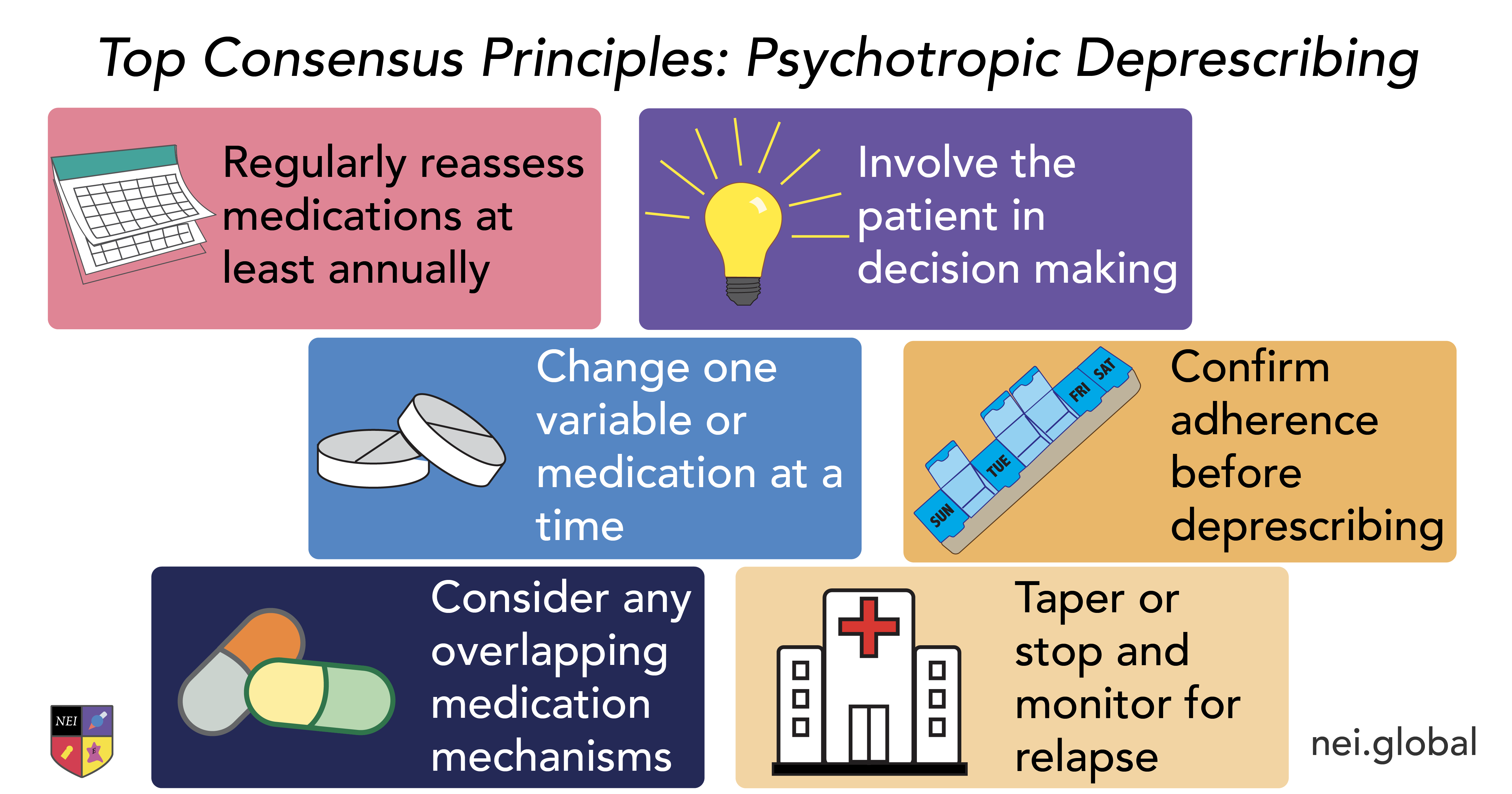
These recommendations reinforce that deprescribing is not simply stopping medication, but a deliberate patient-centered process. For clinicians, this provides a practical framework: regularly reassess the role of each medication, ensure it targets a meaningful symptom, and engage patients in shared decisions about continuation vs discontinuation. Overall, as polypharmacy becomes increasingly common, structured deprescribing may be just as important as prescribing itself.
Reference:
Goldberg JF et al. JAMA Netw Open. 2026;9(2):e260043. Abstract
Additional Education and Practice Resources

NEI Clinical Toolkit
Switching and Deprescribing
Discover treatment guidance, and clinical insights—all designed to simplify decisions and improve patient outcomes.