Most patients with major depressive disorder (MDD) do not achieve full remission with a first-line antidepressant. Data from the landmark STAR*D trial — revisited in a 2023 re-analysis — indicate that the cumulative remission rate across four sequential treatment steps was only 35%. The downstream consequences of non-remission are substantial: higher recurrence risk, chronicity, medical and psychiatric comorbidity, suicidality, excess healthcare utilization, and profound patient dissatisfaction.
Guidelines internationally and in the United States recognize adjunctive antipsychotic therapy as a standard next-step strategy when an antidepressant produces an inadequate response. Currently, five agents carry FDA approval for this indication: aripiprazole, brexpiprazole, cariprazine, quetiapine XR, and lumateperone. Despite their shared label territory, these agents differ meaningfully in pharmacology, receptor binding, efficacy, and tolerability — differences that have rarely been directly compared.
Study Design and Methods
McIntyre and colleagues conducted a pre-registered systematic review and network meta-analysis searching PubMed/MEDLINE, PsycINFO, Cochrane, Embase, and clinical trial registries through July 15, 2025. Only randomized, double-blind, placebo-controlled trials (RCT) enrolling adults (≥18 years) with MDD and documented inadequate antidepressant response were included.
Key Inclusion Criteria
- Agents evaluated: aripiprazole, brexpiprazole, cariprazine, quetiapine XR, lumateperone
- All studies double-blind, placebo-controlled, conducted in outpatient settings
- Primary endpoint: week 6 (per FDA regulatory guidance)
- 22 studies ultimately included, comprising 10,962 participants
Outcomes Assessed
- Co-primary: Efficacy (response rate, ≥50% MADRS reduction) and Acceptability (all-cause discontinuation rate)
- Secondary: Remission (MADRS ≤8 or ≤10), depressive symptom severity reduction (MADRS mean difference), and discontinuation due to adverse events
Key Findings
Efficacy (Response Rate)
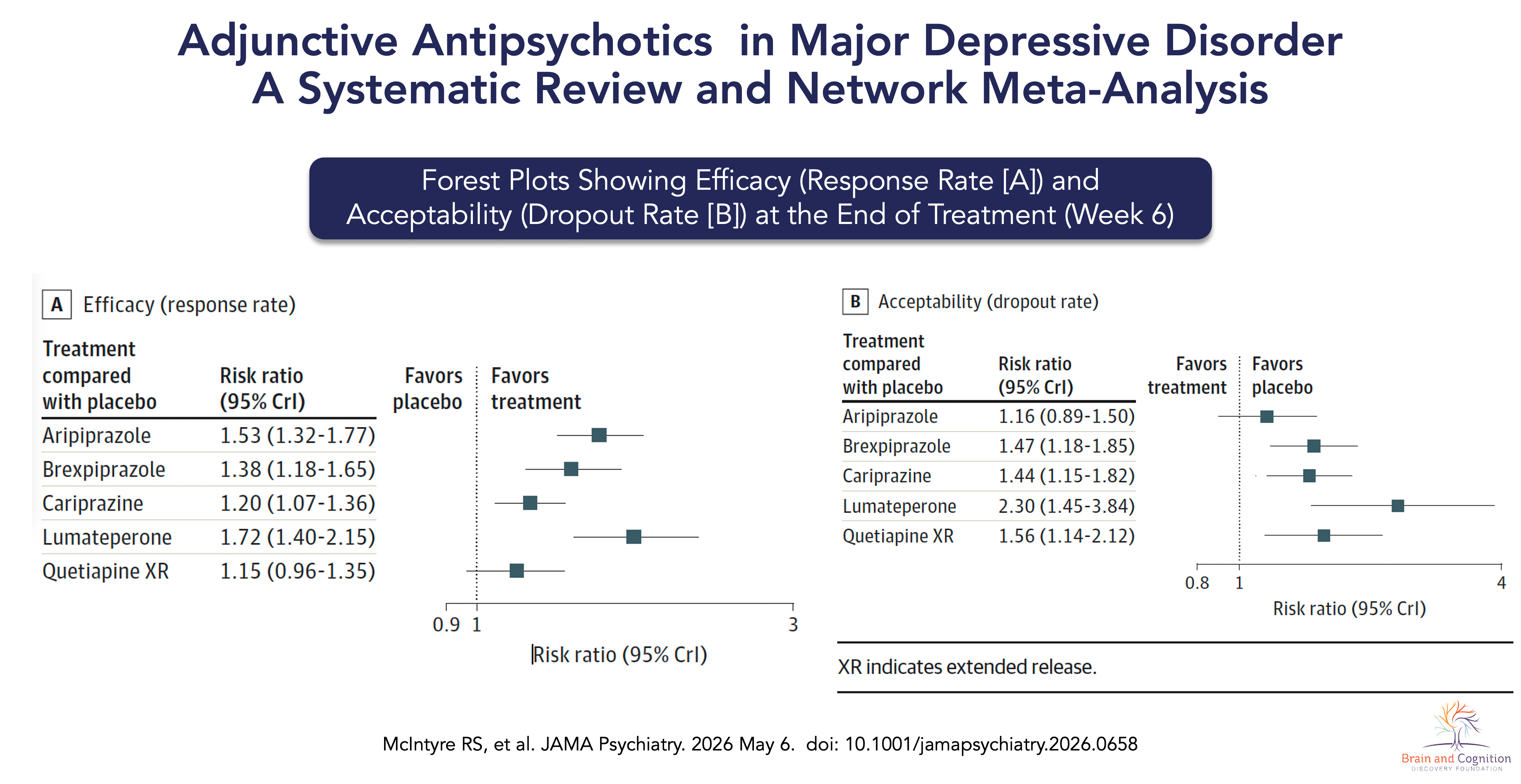
Compared with placebo, all five agents significantly improved response rates, with a clear hierarchy emerging across agents. Lumateperone had the highest efficacy (RR 1.72; 95% CrI 1.40–2.15), followed by aripiprazole (RR 1.53; 95% CrI 1.32–1.77), brexpiprazole (RR 1.38; 95% CrI 1.18–1.65), cariprazine (RR 1.20; 95% CrI 1.07–1.36), and quetiapine XR (RR 1.15; 95% Crl 0.96–1.35). Dose-stratified analyses reinforced this ordering, with lumateperone 42 mg (RR 1.73) and aripiprazole 11 mg (RR 1.67) driving the largest effects at specific doses.
Remission and Symptom Severity
Lumateperone demonstrated the largest effect on remission (RR 1.87; 95% CrI 1.31–2.69) and the greatest reduction in MADRS total score (MD -4.83; 95% CrI -6.42 to -3.21), followed by aripiprazole (RR 1.58 [95% CrI 1.28–1.95] for remission; MD -2.34 [95% CrI -3.33 to -1.37). These findings were consistent across Bayesian and Frequentist analyses and across sensitivity analyses including older adults and studies published from 2010 onward.
Acceptability (All-Cause Dropout)
Acceptability — reflecting real-world treatment persistence — followed a different hierarchy. Aripiprazole had the highest acceptibility (RR 1.16; 95% CrI 0.89–1.50), followed by cariprazine (RR 1.44; 95% CrI 1.15–1.82), brexpiprazole (RR 1.47; 95% CrI 1.18–1.85), quetiapine XR (RR 1.56; 95% CrI 1.14–2.12), and lumateperone (RR 2.30; 95% Crl 1.45–3.84).
Discontinuation Due to Adverse Events
Lumateperone had a relatively higher rate of discontinuation due to adverse events (RR 16.5; 95% CrI 4.57–80.5), followed by quetiapine XR (RR 6.70; 95% CrI 2.90–17.90).
Clinical Implications for Practice
This network meta-analysis provides the most comprehensive comparison of adjunctive antipsychotics in MDD published to date. No single agent dominates across all outcomes. Lumateperone led on efficacy and remission metrics, followed by aripiprazole. Aripiprazole exhibited the highest acceptability, followed by cariprazine. Clinicians must weigh these factors in the context of individual patient priorities, baseline symptom burden, and tolerance for specific adverse effects.
Fewer than 25% of eligible candidates with MDD and inadequate antidepressant response are currently receiving adjunctive antipsychotic therapy. Concerns about 'antipsychotic' stigma and tolerability, limited clinical experience, and access/affordability may be key drivers of this. This evidence base should strengthen clinician confidence in recommending these agents earlier and more consistently.
Some Limitations to Consider
- This network meta-analysis is confined to acute (6–8 week) endpoints
- Results cannot be generalized to agents that were not included
- Findings may not generalize to real-world patients who often would not qualify for RCT enrollment
- The definition of 'inadequate response' varied across agent-specific trial programs
- Moderators such as anxious distress, mixed features, anhedonia, and trauma history were not evaluated
- Magnitude of placebo response across the included studies was highly variable
Reference: McIntyre RS, Stahl SM, Shim SR et al. Adjunctive Antipsychotics in Major Depressive Disorder: A Systematic Review and Network Meta-Analysis. JAMA Psychiatry 2026; doi: 10.1001/jamapsychiatry.2026.0658 Abstract
Additional Education and Resources: